Given its impact on employees’ well-being, and their ability to function both at work and socially, providing access to efficient treatment options early can have a significantly positive impact

This article was produced in partnership with Novartis Pharmaceutical Canada Inc.
Hidradenitis Suppurativa (HS), also known as acne inversa, is a misunderstood, and often misdiagnosed, chronic inflammatory skin condition affecting approximately 1% of Canadians.1 The first symptoms of HS start to appear between puberty and before the age of 40, and is three times more common in women than men, overshadowing a unique time of transition into adulthood and hindering the most active years of their lives.2
Living with Hydradenitis Suppurativa
HS is characterized by painful inflamed bumps called “nodules” that can drain fluid or foul-smelling pus (abscesses). The nodules can eventually heal but may recur at any time and can lead to irreversible scarring and the formation of tunnels under the skin known as “fistulas”. HS often manifests in cycles of unpredictable, intense flare-ups and periods when the skin is clear.
During a flare-up, painful and swollen lesions tend to develop in several problematic areas where skin rubs together such as the armpits, groin, buttocks and breasts.3 In addition to causing chronic pain, fistulas can restrict range of motion in the limbs when accompanied by significant scarring. The severity of the disease is commonly assessed using the Hurley staging system; mild disease represents approximately 44% of cases, whereas moderate and severe disease represent 44% and 12% of HS cases, respectively.4
The chronic and recurrent nodules, abscesses, tunnels, scaring, pain and itching all contribute to the burden of HS and significantly hinder the person’s quality of life (QoL).6 Symptoms of HS can be physically debilitating for individuals as they experience impairment of activities and daily living, leading to greater reliance on caregivers and a loss of independence. Studies have shown that symptoms of HS, such as bad-smelling discharge and disfiguring scars, and the lack of disease control can negatively impact emotional and social functioning.7 Additionally, the location of the lesions is often involved in sexual intimacy, which negatively impacts sexual health and quality of life while increasing psychological comorbidities.8
Most individuals with HS live with recurring symptoms over many years, which can lead to chronic pain, reduced mobility due to significant scarring and poor overall QoL. Although early recognition and prompt access to treatment has been shown to reduce the burden of HS, many individuals endure debilitating symptoms for years without access to effective treatment. Most notably, a survey conducted by the Canadian Skin Patient Alliance (CSPA) in 2020 showed that the average time between symptom onset and HS diagnosis was seven years and that participants were on average misdiagnosed three times before receiving their correct HS diagnosis.9
Social stigma around the disease can amplify the fear of judgement and exacerbate feelings of isolation. Given their experiences, many individuals with HS ultimately suffer from isolation, anxiety, depression, and suicidal ideation. Notably, 43% of patients with HS are diagnosed with depression and with greater risk of death from suicide compared to the general population.10,11
Impact at work for employees with HS
HS usually affects young adults during prime career- and family-building years, with impairment increasing with disease severity. During flare-ups, symptoms can prevent employees from sitting at a desk, walking, lifting, wearing a uniform, etc.
Understandably, participants of the CSPA survey reported missing on average 3 days of work per month and many experienced job loss due to their HS condition. A study conducted in the United States in 2019 revealed that participants with HS had higher total annual indirect costs including disability and medically related absenteeism costs, number of days of work loss and risk of leaving the workforce compared to individuals without HS (Figure 1).9,12
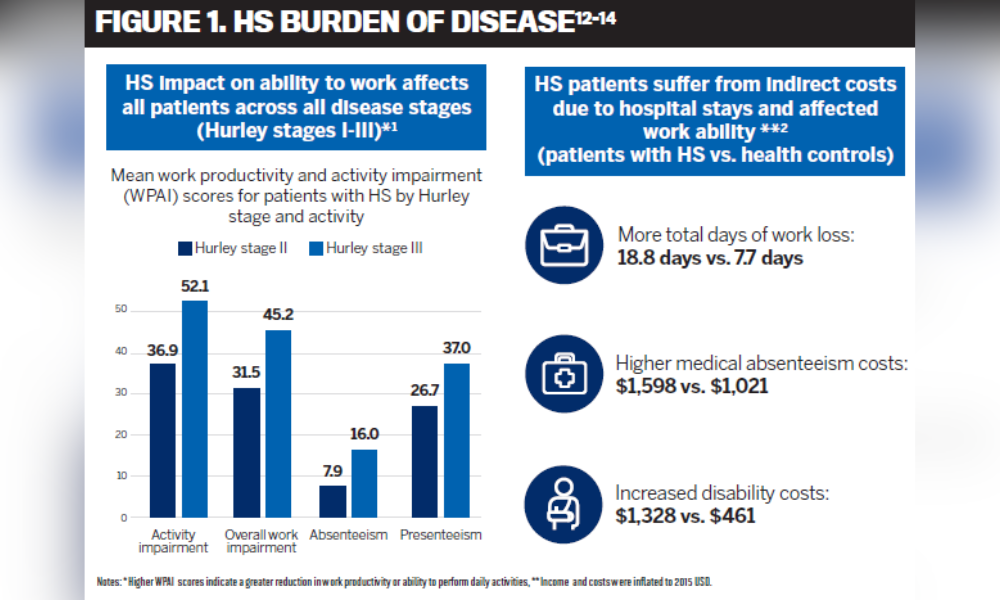
HS can also negatively impact employees’ ability to be productive at work as shown in a recent Danish study where only 57% of participants with HS were employed, of which over 21% reported missing work and 60% reported loss of work productivity during the previous week as a result of HS. Notably, overall work productivity was reduced by 27% and impairment was shown to increase with disease severity.13
The ramifications of the disease can also extend into their social lives, impeding their ability to engage with their significant others and family members and socialize with friends and colleagues.9
What are the treatment options?
The goal of currently available therapies is to reduce symptoms, alleviate pain, heal existing lesions, treat infections, prevent subsequent flare-ups and stop disease progression. In addition to lifestyle changes and pain medication, HS treatment options include topical therapies, antibiotics, hormone therapy, steroids, biologics, laser therapy and surgery.
Most individuals with HS require lifestyle, medical and/or surgical interventions to effectively manage their symptoms. However, each individual responds differently to any given therapy and HS patients often have other medical conditions which can make developing an effective treatment plan very challenging.5 This was demonstrated in the CSPA survey where more than 60% of the participants reported dissatisfaction with the treatment options, after trying an average of 15 different therapies.9 Further, a separate report based on interviews with 12 patients and 16 healthcare providers (HCPs) conducted in 2022 revealed 16 areas of unmet needs, which were either related to treatment outcomes or the care process, and 13 important treatment attributes. The most frequently reported unmet needs by patients and HCPs were lacking quality-of life improvement, low treatment effectiveness and inadequate pain control.15
Antibiotics and surgery can be effective therapies in some HS patients but have limiting factors such as limited short-term response, development of bacterial resistance, painful recovery and permanent scarring.5,16,17 With suboptimal treatments, HS can progress to more severe stages.5 Early treatment with biologics has been shown to delay disease progression and can prevent the severe form of the disease such as scaring (Figure 2).18
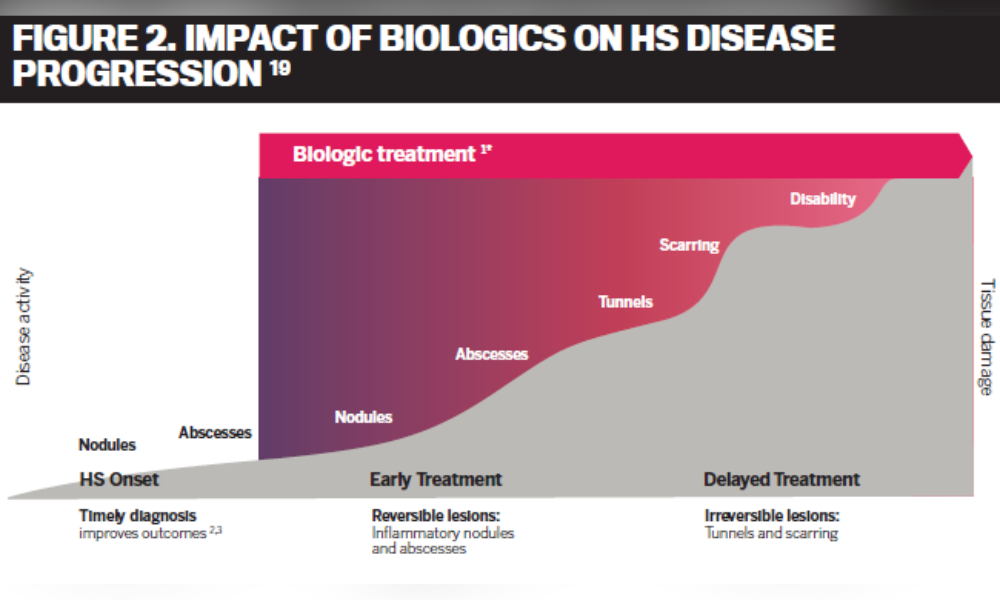
HS patients will benefit from therapies with different mechanisms of action to allow for more personalized treatment plans. Currently, two biologic drugs – adalimumab (HUMIRA® and biosimilars) and COSENTYX® (secukinumab) – are approved by Health Canada for the treatment of moderate to severe HS. Adalimumab is a recombinant human immunoglobulin (IgG1) monoclonal antibody which works by blocking tumor necrosis factor alpha (TNFα).20 COSENTYX®, which has been marketed in Canada for other conditions since 2015, was recently approved by Health Canada for the treatment of HS. COSENTYX® works differently via a new mechanism of action by selectively neutralizing the body’s interleukins (IL)-17A.21,22
What does this mean for plan sponsors?
Despite the abundance of images and information about the disease in the public domain, HS is a relatively obscure skin condition for many people, including some health care providers, leading to delays in treatment start and misdiagnosis, resulting in irreversible lesions and prolonged disease burden. Access to adequate treatments can significantly reduce indirect costs due to absenteeism, presenteeism, productivity loss and disability costs.
Although there is a tremendous unmet need in HS, only a small proportion of plan members have access to new therapies that are safe, effective in limiting disease progression and have been shown to be successful treatment options for long-term use.9 One way to improve health outcomes would be to ensure that physicians have timely access to the full range of approved medications with different mechanisms of action, including newly approved therapies, in order to effectively manage each patient with HS. Plan sponsors should ask their benefits providers about their approach in reviewing new therapies and how comprehensively their drug plan is maintained to support employees with HS.
Given the impact of HS on employees’ well-being, and their ability to function both at work and socially, providing access to safe and efficient treatment options early can have a significantly positive impact on employees, help them remain productive at work and enable them to engage in healthy and rewarding experiences in their personal lives.
References:
1 Kimball et al. (2023), 2Alikhan et al. (2009), 3Canadian Hidradenitis Suppurative Foundation, 4 Canadian market research study, Novartis data on file (2022), 5 Johnston et al. (2022), 6 Chernyshov et al. (2019), 7 Alavi et al. (2018-MR), 8 Alavi et al. (2018-JN), 9 Canadian Skin Patient Alliance, 10 Thorlacius et al. (2018), 11 Vazquez et al. (2013), 12 Tzellos et al. (2019), 13 Yao et al. (2020), 14 Kimball et al. (2020), 15 Willems et al (2022), 16 Fischer et al. (2017), 17 Burney (2017), 18 Alikhan et al. (2019), 19 Martorell et al. (2016), 20 Abbvie, HUMIRA Product Monograph (2022), 21 Novartis, COSENTYX Product Monograph (2023), 22 Skroza et al. (2017).



